For example, water consists of one oxygen atom covalently bonded to two hydrogen atoms (H 2O). Elements will share a pair of electrons in order to gain a complete outer shell and a stable electron configuration. Covalent CompoundsĬovalent compounds are chemical compounds that are formed by the sharing of electron pairs between elements. What kinds of bond two elements enter into is determined by those elements electronegativities - a measure of how greatly those elements attract electrons. There are 3 major kinds of chemical compounds, each differentiated by how the constituent atoms are held together covalent, ionic, and metallic. Types Of Chemical Compounds With Examples Chemical compounds always have the same proportions of constituents every molecule of salt is composed of one sodium and one chlorine atom (NaCl). Table salt (NaCl) is a compound because it is composed of a sodium atom (Na) and a chlorine atom (Cl) that are bonded together. A sample of steel could have more carbon or less iron in it.Ĭhemical compounds, on the other hand, consist of substances that share chemical bonds with each other. Moreover, the proportion of constituents of steel can differ. Steel is composed of quantities of iron (Fe) and carbon (C), but those elements are not chemically bonded. A mixture might include a number of different elements, but in a mixture, none of the elements share chemical bonds with each other. Mixture: Examples of Chemical SubstancesĪ chemical compound is different than a mixture. There are three major types of chemical compounds, each defined by how their atoms are bonded together: covalent compounds, ionic compounds, and metallic compounds. The main mechanism that drives the bonding of chemical compounds is the share or transfer of valence electrons. “Modern chemistry, with its far-reaching generalizations and hypotheses, is a fine example of how far the human mind can go in exploring the unknown beyond the limits of human senses.” - Horace G. Compounds interact through chemical reactions, during which atomic bonds are broken and reform into different compounds. Every chemical compound has a chemical formula, a notation that describes the relative proportions of elements that constitute a given compound. So I'm feeling veryĬonfident just eyeballing it, just estimating, this is going to be a little bit, have an average atomic massĪ little bit less than 88, which tells me that this is strontium.A chemical compound is, for example, a chemical substance that is formed by the bonding of two or more different chemical elements. To do the calculation, we could feel confident we're not going to be as low as rubidium. It's exactly what we predicted, a little bit less than 88, and rubidium is a lot less than 88. What element here has an atomic mass a little bit less than 88? Well, yttrium is 88.91, but we know it can't be thatīecause none of the isotopes have an atomic mass above 88. So our average atomic mass is going to be a little bit less than 88. But these other isotopes, these other versions of the element that have a different number of neutrons, which changes its atomic mass, they're going to bring the average down. When we're taking the weighted average, we have the highest Going to be close to 88 because that's where the Periodic table of elements, or we could just try to estimate it. We could type this into a calculator and get some number and then look that up on a
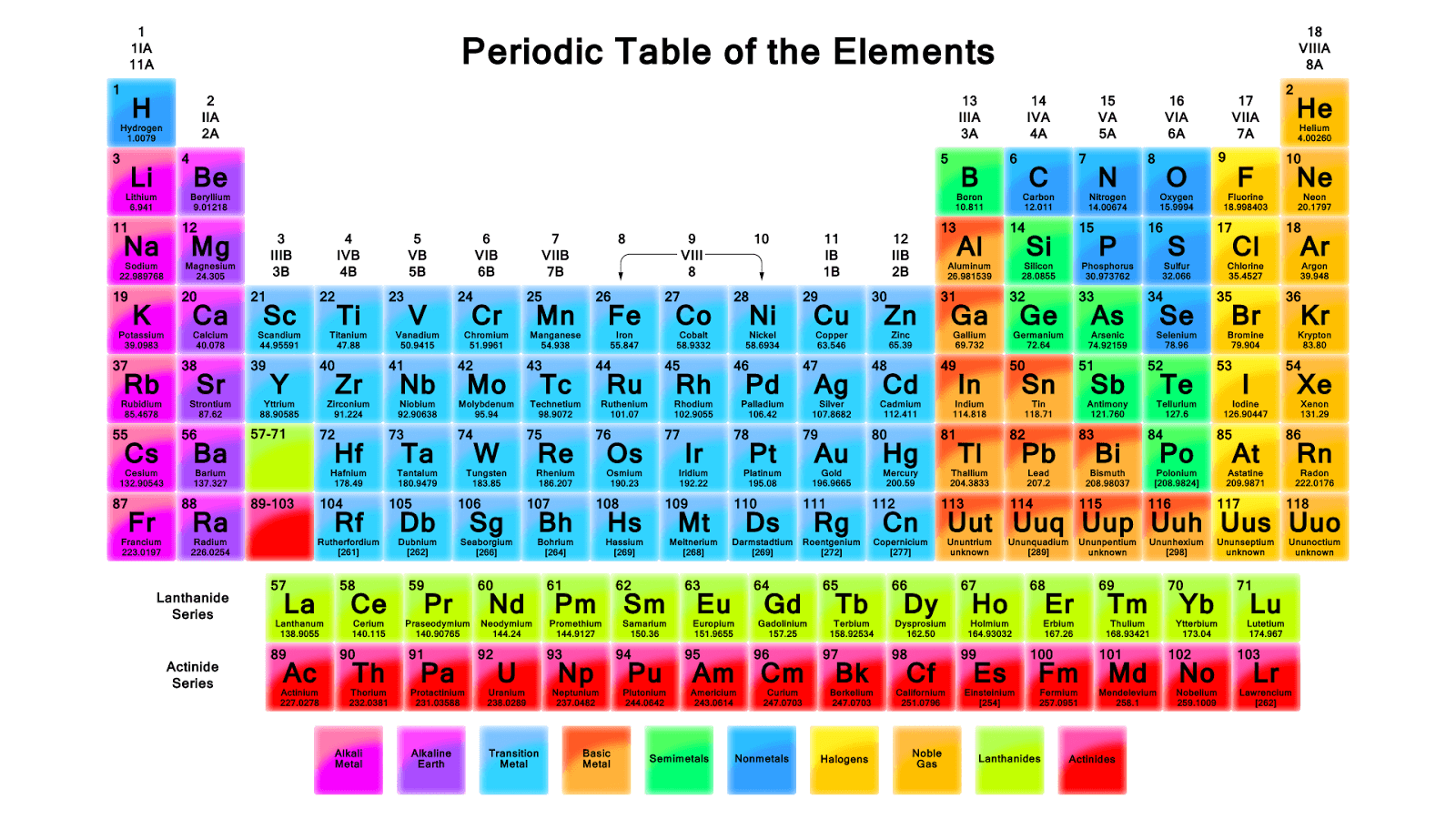
And so if we were to do this calculation, this is our estimate of the average atomic This is 89, and then this gets us to 99, so then another 1%, 0.01 times 84.
Examples of elements plus#
We could calculate it as 0.82 times 88, plus, let's call this 7%, so 0.07 times 87, plus 10%, 0.1, times 86, plus, let's see, it should add up to 100%. The average atomic mass of this mystery element is. And so from this information, we can try to estimate what It looks like 10% has an atomic mass of 86 universal atomic mass units, and it looks like about 1% of our sample has an atomic mass of 84 About, this looks likeĪbout 7% of our sample has an atomic mass of 87 So what this is telling us is, this looks like maybe, I don't know, let's call this 82% of our sample has an atomic mass of 88
/Argon-58e6aa855f9b58ef7e08cda8.jpg)
Spectrum for an average sample of a pure element is shown below. Take a sample of a substance and think about the various atomic masses of the different isotopes Known as mass spectrometry or mass spectroscopy. Well, scientists have a method, and we go into the details, or more details, in other videos, called mass, sometimes it's So let's say that we have some mystery substance here, and we know that it's a pure element, and we need to figure out what it is.
